Toisesta ketjusta bongattu treidaajien työtä helpottava palvelu saattaisi antaa vihjeitä FDA-prosessista, joka taitaa olla vasta aluillaan.
https://www.fdatracker.com/fda-calendar/
Se on kokonaisuutena trial- ja patenttiseurantoineen kk-maksullinen, mutta ilmainen näkymä on FDA-kalenteriin, jossa ei Faronia nyt näkynyt.
”This calendar tracks upcoming PDUFA drug approval dates and FDA advisory committee meetings.”
FDA-prosessista
https://www.fda.gov/media/72253/download
Type B meetings should be scheduled to occur within 60 days of FDA receipt of the written meeting request.
If a meeting request is granted, CBER or CDER will notify the sponsor or applicant of the decision and schedule the meeting by determining the meeting type, date, time, length, place, and expected FDA participants.
A meeting package should be submitted to the appropriate review division so that it is received in accordance with the following time frames:
• Type B meeting — At least 4 weeks before the formal meeting.
CBER and CDER hold internal meetings to discuss meeting packages and to gain internal agreement on the preliminary responses to a sponsor’s or applicant’s questions. We may communicate these preliminary responses to the sponsor or applicant.
Before the end of the meeting, FDA attendees and the sponsor or applicant attendees should summarize the important discussion points, agreements, clarifications, and action items. Generally, the sponsor or applicant will be asked to present the summary to ensure that there is mutual understanding of meeting outcomes and actions.
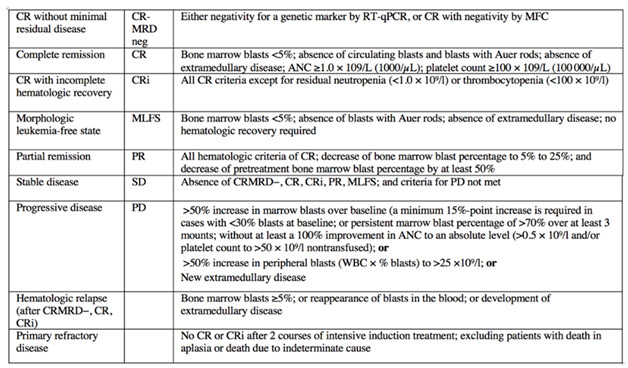
Tässä on monta vaihetta, josta Faron voisi tiedottaa, mutta onko mikä RNS:n arvoinen? 1) Tapaamispyynnön lähetys 2) Tapaamisen myöntö ja tapaamisen aikataulu 3) Paketin lähetys 4) Lisäpyynnöt pakettiin liittyen 5) Tapaamisen tulos